You are here
Content
Endothelial cells display marked organ-specific heterogeneity. Endothelial heterogeneity mediates organ-specific differentiation of the microvascular niche comprising various other cell populations besides endothelial cells such as vascular wall cells (pericytes), parenchymal and stromal cells, but also stem cells. Organ-specific differentiation of the microvascular niche is induced by so-called angiokines. Angiokines are cytokines that are produced by endothelial cells and display an organ-specific pattern of secretion.
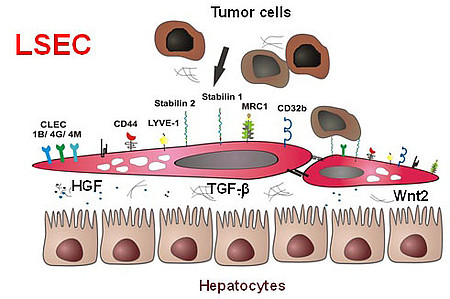
The microvascular bed of the liver represents a prime example of the organ-specific heterogeneity of the microvascular niche. Here, the organ-specific microvascular endothelial cells are called liver sinusoidal endothelial cells (LSEC). LSEC differ from normal endothelial cells by their special morphology including lack of a basement membrane. Pathological changes of these highly specialized, discontinuous liver sinusoids contribute to severe liver diseases such as alcoholic steatohepatitis and liver cirrhosis. During hepatocarcinogenesis and metastasis to the liver, LSEC trans-differentiate towards a capillary phenotype (capillarization). Normal LSEC display a high capacity of endocytosis for clearance of noxious factors from the peripheral blood. To this end they have a complex cytoskeleton with sieve plates and fenestrations and they express a special set of endocytic receptors. LSEC are also involved in regulating portal blood pressure.
In the recent years, our group has been able to identify several novel sinusoidal endothelial proteins using classical biochemical methods as well as novel molecular approaches including gene expression profiling. Stabilin1 (Stab1) and Stabilin2 (Stab2) were first identified as novel marker proteins of discontinuous sinusoidal endothelial cells; further bioinformational analysis was used to classify them as endocytic scavenger receptors. In addition, we could show that the stabilins considerably contribute to the clearance function of LSEC. As ligands for the stabilins, we identified a wide range of molecules, i.e. hyaluronic acid, AGE-modified proteins, the extracellular matrix protein SPARC and others such as chitiniase-like proteins (SICLP), cytokines (GDF15), and hormones (placental lactogen).
In order to analyze the scavenger functions of LSEC in more detail, Stab1- and Stab2-deficient mice were generated. While Stab1 and Stab2 single knockout mice were grossly normal, mice deficient for both stabilins dye early and suffer from severe hepatic and renal fibrosis. In complicated animal and transplantation experiments, we could show that glomerulofibrotic nephropathy of stab1/2-double deficient mice is caused by impaired clearance of noxious blood substances from the peripheral circulation by LSEC.
In our group, we focus on four lines of experiments to further investigate the basic mechanisms and functions of microvascular heterogeneity in the liver:
- Identify and characterize the mechanisms that regulate and control LSEC differentiation during development and in the adult organism in health and disease
- Identify and characterize novel ligands of Stab1/2 and analyze Stabilin-mediated clearance processes in metabolic diseases
- Functionally analyze novel LSEC-specific transcription factors and other unknown molecules such as Gata4 and Leda1
- Define the role of endothelial heterogeneity and of the organ-specific cellular and molecular interactions within the hepatic vascular niche during metastasis
Project-related publications
- Géraud C, Koch PS, Damm F, Schledzewski K, Goerdt S. The metastatic cycle: metastatic niches and cancer cell dissemination. J Dtsch Dermatol Ges, 12: 1012-9, 2014.
- Géraud C, Koch PS, Goerdt S. Vascular niches: endothelial cells as tissue- and site-specific multifunctional team players in health and disease. J Dtsch Dermatol Ges, 12: 685-9, 2014.
- Géraud C*, Mogler C*, Runge A, Evdokimov K, Lu S, Schledzewski K, Arnold B, Hämmerling G, Koch P, Breuhahn K, Longerich T, Marx A, Weiss C, Damm F, Schmieder A, Schirmacher P, Augustin HG, Goerdt S. Endothelial transdifferentiation in hepatocellular carcinoma: loss of Stabilin-2 expression in peri-tumorous liver correlates with increased survival. Liver International, 33: 1428-40, 2013.
- Evdokimov K, Biswas S, Adrian M, Weber J, Schledzewski K, Winkler M, Goerdt S, Géraud C. Proteolytic cleavage of LEDA-1/PIANP by furin-like proprotein convertases precedes its plasma membrane localization. Biochem Biophys Res Commun, 434: 22-27, 2013.
- Géraud C*, Evdokimov K*, Straub BK, Peitsch WK, Demory A, Dörflinger Y, Schledzewski K, Schmieder A, Schemmer P, Augustin HG, Schirmacher P, Goerdt S. Unique cell type-specific junctional complexes in vascular endothelium of human and rat liver sinusoids. PLoS ONE, 7:e34206, 2012.
- Schledzewski K*, Géraud C*, Arnold B, Wang S, Gröne HJ, Kempf T, Wollert KC, Straub BK, Schirmacher P, Demory A, Schönhaber H, Gratchev A, Dietz L, Thierse HJ, Kzhyshkowska J, Goerdt S. Deficiency of liver sinusoidal scavenger receptors stabilin-1/-2 in mice causes glomerulofibrotic nephropathy via impaired hepatic clearance of noxious blood factors. Journal of Clinical Investigation, 121:703–714, 2011.
- Géraud C*, Schledzewski K*, Demory A, Klein D, Kaus M, Peyre F, Sticht C, Evdokimov K, Lu S, Schmieder A, Goerdt S. Liver sinusoidal endothelium: a microenvironment-dependent differentiation program in rat including the novel junctional protein liver endothelial differentiation-associated protein-1. Hepatology, 52: p. 313-26, 2010.
Context Column
Contact

PD Dr. Cyrill Géraud
Arbeitsgruppe Mikrovaskuläre Heterogenität
Department of Dermatology, Venereology and Allergology
Medical Faculty Mannheim
Heidelberg University
Theodor-Kutzer-Ufer 1-3
68167 Mannheim
Phone +49 621 383-2617
cyrill.geraud@umm.de